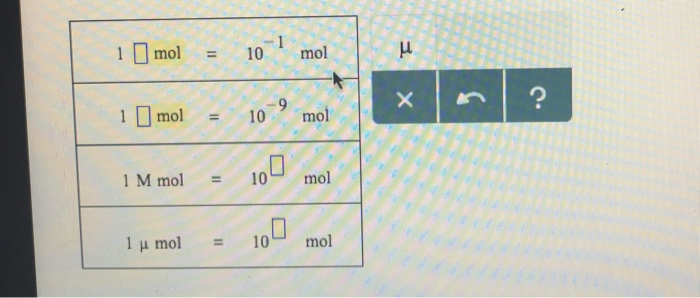
SOLVED: a mole of sodium atoms contains 6.02 x 10^23 atoms. How many moles would be needed in order to have 2.50 x 10^24 atoms?

Bonide MOLEMAX Mole & Vole Repellent Granules, 10 lbs. Ready-to-Use, Outdoor Lawn & Garden Mole Control, People & Pet Safe
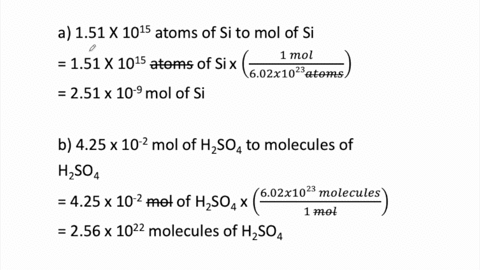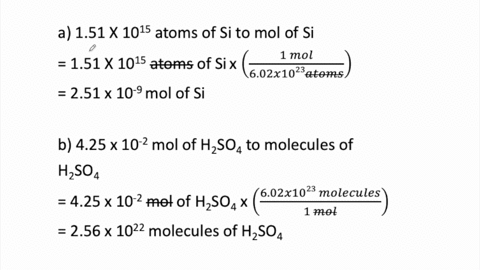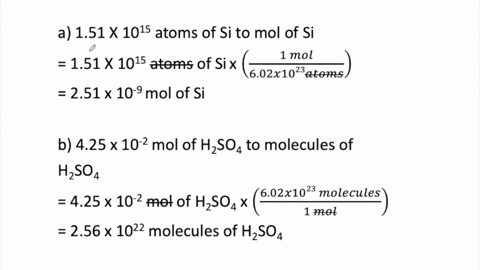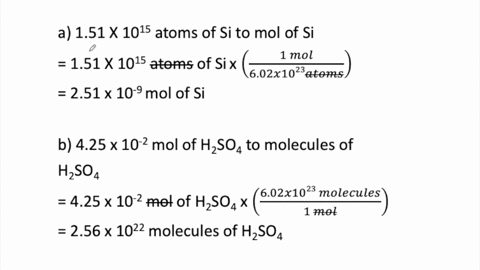
SOLVED:Determine the number of moles in each substance. a. 3.25 ×10^20 atoms of lead b. 4.96 ×10^24 molecules of glucose c. 1.56 ×10^23 formula units of sodium hydroxide d. 1.25 ×10^25 copper (II) ions
4 A-3 The Mole The mole (abbreviated mol) is the SI unit for the amount of a chemical species. It is always associated with a ch
![Solubility product of AgCl is 1.8 × 10^-10 . Calculate its molar solubility and solubility in g/L. [Molar mass of AgCl is 143.5 g mol^-1 ] Solubility product of AgCl is 1.8 × 10^-10 . Calculate its molar solubility and solubility in g/L. [Molar mass of AgCl is 143.5 g mol^-1 ]](https://dwes9vv9u0550.cloudfront.net/images/5064616/add4819a-6281-4659-ab5c-8471f2633afa.jpg)
Solubility product of AgCl is 1.8 × 10^-10 . Calculate its molar solubility and solubility in g/L. [Molar mass of AgCl is 143.5 g mol^-1 ]

If `NaCl` is doped with `10^(-3)` mol% of `SrCl_(2)`, what is the concentration of cation vacanc... - YouTube

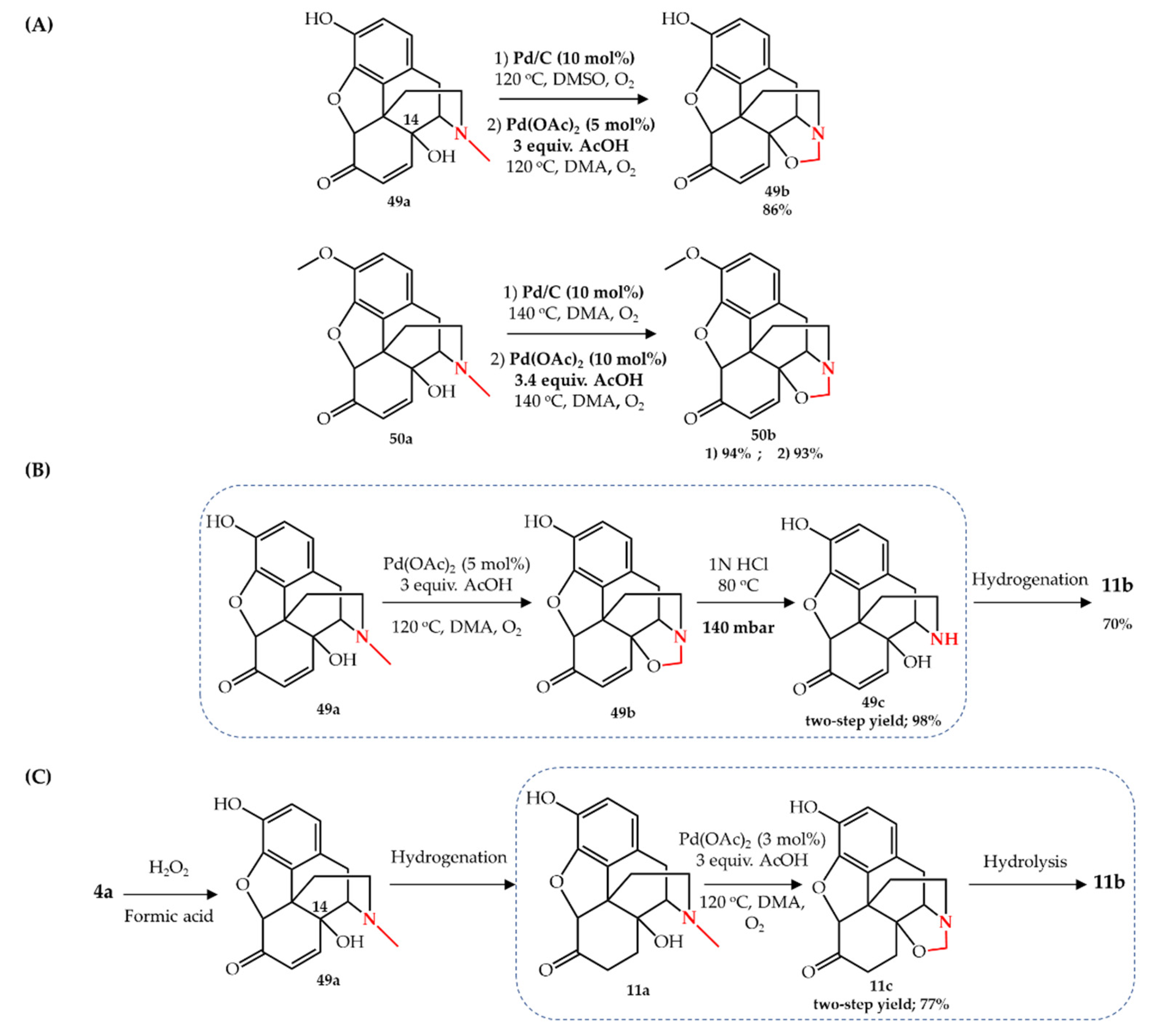
Oxidation of secondary alcohols using 10 mol% lanthanum triflate as... | Download Scientific Diagram

How many atoms or molecules are present in 1.0 mol of Au? How am I able to find the answer? | Socratic